
Testing
Skin care
KEYWORDS
peer-reviewed
Proving Benefits of Microbiome Skin Care
Christiane Uhl
Head of Sales Department (Domestic & Austrian market), Trainings & Special Projects,
Courage + Khazaka electronic GmbH, Cologne, Germany
ABSTRACT: The aim of this article is to discuss ways of testing the benefit of cosmetics with a microbiome claim. Research on the skin microbiome has shifted from focusing solely on pathogens toward recognizing the beneficial role of commensal microorganisms. This paradigm change has sparked strong interest in microbiome tending skin care, yet scientific proof of efficacy remains challenging. Microbiome analyses can demonstrate shifts in microbial composition after product use, but high inter- and intra-individual variability prevents the definition of an “ideal” microbiome. Classical, non-invasive biophysical methods therefore remain essential for substantiating product claims. Measurements of barrier integrity, hydration, pH, sebum, irritation etc. are reliable, reproducible, and directly linked to skin health.
??????????????????
“
“A study in healthy women providing probiotic yogurt for four weeks showed an improvement in emotional responses as measured by brain scans”
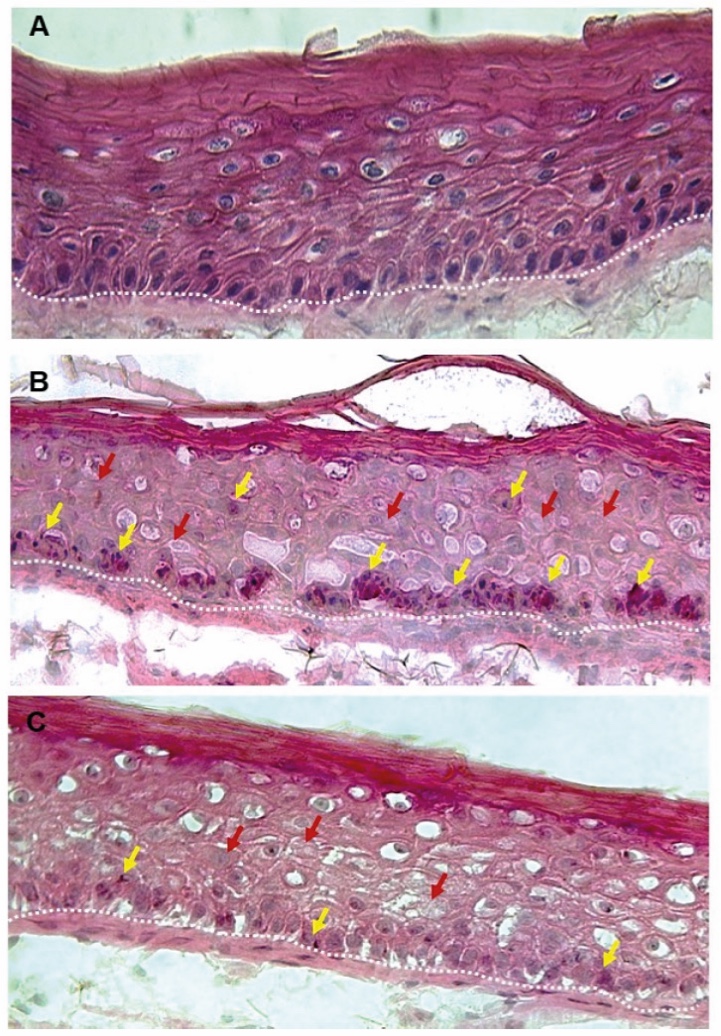
Figure 1. Skin Section with Microbiome. Most microorganisms live in the superficial layers of the stratum corneum and in the upper parts of the hair follicles. Some reside in the deeper areas of the hair follicles and are beyond the reach of ordinary disinfection procedures. There bacteria are a reservoir for recolonization after the surface bacteria are removed.
Materials and methods
Studies of major depressive disorder have been correlated with reduced Lactobacillus and Bifidobacteria and symptom severity has been correlated to changes in Firmicutes, Actinobacteria, and Bacteriodes. Gut microbiota that contain more butyrate producers have been correlated with improved quality of life (1).
A study in healthy women providing probiotic yogurt for four weeks showed an improvement in emotional responses as measured by brain scans (2). A subsequent study by Mohammadi et al. (3) investigated the impacts of probiotic yogurt and probiotic capsules over 6 weeks and found a significant improvement in depression-anxiety-stress scores in subjects taking the specific strains of probiotics contained in the yogurt or capsules. Other studies with probiotics have indicated improvements in depression scores, anxiety, postpartum depression and mood rating in an elderly population (4-7).
Other studies have indicated a benefit of probiotic supplementation in alleviating symptoms of stress. In particular, researchers have looked at stress in students as they prepared for exams, while also evaluating other health indicators such as flu and cold symptoms (1). In healthy people, there is an indication that probiotic supplementation may help to maintain memory function under conditions of acute stress.
Research into microorganisms has a long history, but for much of the 20th century the focus was almost exclusively on pathogens—microbes responsible for disease—and their elimination. Cleansers, antibiotics, and disinfectants were studied and promoted primarily for their ability to eradicate harmful organisms. Only in recent decades has science begun to explore the beneficial role of commensals. The results of the Human Microbiome Project (1) coupled with advances in metagenomics have triggered remarkable momentum in microbiome research, highlighting the living body not only as a host to pathogens but also as a complex ecosystem (Figure 1) whose balance is critical for health.
Figure 1. The skin is a natural habitat for a rich variety of microorganisms.
The connection between microbiome and body health requires little introduction. Numerous scientific publications have detailed how our microbial cohabitants influence immunity and well-being (2).This new perspective has accelerated interest in the skin microbiome as both a potential cause of skin problems and a possible key to their solution (3). Still, despite intense research, consensus on what constitutes a “healthy” microbiome remains elusive.
Several factors contribute to this uncertainty. Microbiome composition varies not only between individuals—shaped by genetics, health, nutrition, and environment—but also across body sites on the same person (4),(5), (6), (7). The skin of the forehead, armpit, and forearm, for example, all host distinct microbial communities. This heterogeneity makes it difficult to define universal standards.
Classic microbiological claims are relatively straightforward to prove. The germ-killing action of soaps, antibiotics, or disinfectants can be demonstrated by traditional methods targeting individual species (Figure 2). Similarly, claims that products “leave the microbiome untouched” can be evaluated through metagenomic sequencing.
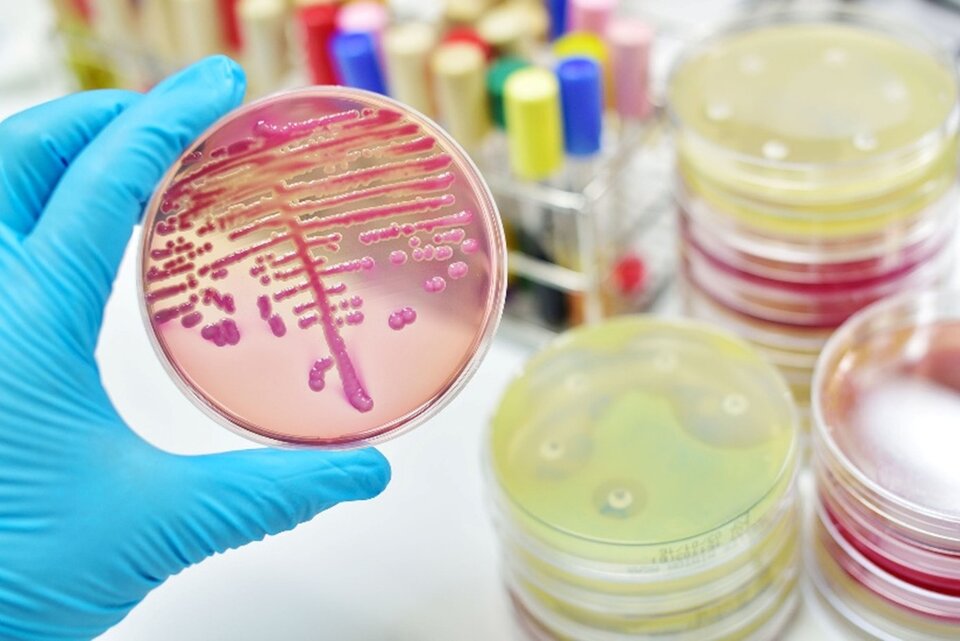
Figure 2. Traditional microbiologic testing methods such as bacterial cultures on agar plates can show the germ-killing activity of substances.
Figure 2. Traditional microbiologic testing methods such as bacterial cultures on agar plates can show the germ-killing activity of substances. |
For chronic skin conditions such as atopic dermatitis and acne, shifts in microbial composition can serve as indirect evidence of efficacy. Nevertheless, translating microbiome data into clear statements about product benefits for healthy skin remains nearly impossible. There is no single global guideline specifically for proving microbiome cosmetic claims, but companies must adhere to existing cosmetic regulations such as the EU Cosmetics Regulation 1223/2009, ensuring products are safe and deliver on their claims (8). The current scientific consensus is that skin microbiome and skin health are inextricably intertwined: a healthy microbiome provides healthy skin andhealthy skin promotes a balanced microbiome (9) , (10), (11), (12).Aspects of skin health can be and have been measured for years by non-invasive, globally acknowledged biophysical methods.
Established Methods to Assess Skin Health
Skin Barrier Function
The skin’s primary role is to act as a protective barrier between the internal body and the external environment. The strength of this barrier is a key determinant of skin health. One of the most widely used assessments is the measurement of the TransEpidermal Water Loss (TEWL) (Figure 3), which quantifies the amount of water evaporating through the skin into the environment (13). Elevated TEWL indicates impaired barrier integrity, a hallmark of many skin disorders (14), (15), (16). With this straightforward method, even subtle barrier damage can be easily detected.

Figure 3. Tewameter® measurement with open measurement chamber does not influence the natural evaporation from the skin.
Skin Surface Hydration
Stratum corneum hydration refers to the water content of the uppermost layer of skin, which is crucial for the skin barrier and the regulation of important functions. Its water holding capacity depends on the presence of natural moisturizing factors and its barrier function. Since this skin layer interacts strongly with the environment, it is easily affected by external influences such as dry air or frequent washing, but can also be influenced by intrinsic conditions (17). Low hydration results in dry, scaly skin and a compromised barrier through which pathogens may enter (18), (19). Excess hydration, for example from continuous wet exposure, leads to a maceration of corneocytes, again weakening the skin’s structural integrity (20), (21).
Balanced hydration is therefore critical for skin health. Various instruments measure stratum corneum hydration, employing methods such as impedance, capacitance, infrared, near-infrared, or Raman spectroscopy.
Skin pH Value
The skin surface pH plays a fundamental role in buffering acids or alkaline substances and shaping microbial communities. Many beneficial bacteria thrive in the slightly acidic conditions of the healthy skin surface. In atopic dermatitis, elevated pH levels are often observed alongside increased colonization by Staphylococcus aureus (22).
By measuring skin surface pH, researchers can determine whether products support or destabilize the microbiome’s preferred acidic environment.

Figure 4. The occurrance of acne is correlated to excess sebum production.
Sebum Levels
Sebum, secreted in sebaceous gland-rich areas such as the face and scalp, is another crucial determinant of microbial ecology. The role of sebum in atopic dermatitis was often dismissed in the past. More recent studies however, have uncovered that in diseased skin conditions such as atopic dermatitis, sebum levels are considerably lower than in healthy individuals (23). On the other hand, excess sebum production, providing a nutrient source for Cutibacterium acnes, is correlated to the occurrence of acne (Figure 4) (24), (25).
Sebum production can be measured with a Sebumeter® or collected and analysed by microporous tapes absorbing the sebum from the skin surface. Additionally, C. acnes activity can be monitored using specialized imaging: the bacterium produces porphyrins that fluoresce under certain UV wavelengths (Figure 5).
Monitoring both sebum production and bacterial activity provides insights into the effectiveness of anti-acne treatments (26).
Blemish Evaluation
Blemishes, including pimples and blackheads, signal deviations from an optimal skin condition. They can be assessed visually or with high-resolution standardized photography (27). When photography is used, special attention should be paid to ambient light, positioning of the face, and background colours such as clothes and hair that influence camera settings and affect comparability. Dedicated imaging systems with controlled illumination and fixed positioning solve this issue and improve reproducibility, enabling both subjective scoring and automated analysis.
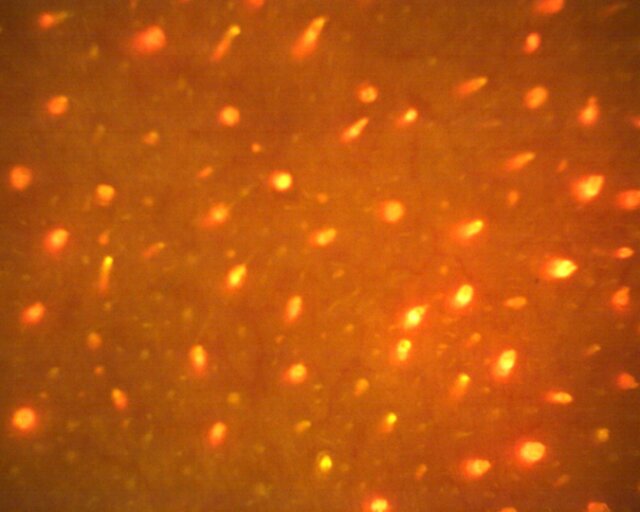
Figure 5. Strong fluorescence of the Cutibacterium acnes porphyrins under UV-illumination of the Visiopor® camera.
Healing Capacity
The skin’s healing ability reflects its resilience and overall health. Healing can be assessed directly, for example by monitoring wound closure after controlled biopsies or excisions. Luckily, also less painful alternatives exist for assessing the healing ability of the skin, especially the barrier regeneration. One method involves standardized repetitive tape stripping (e.g. with Corneofix® or D-Squame®) (Figure 6). This controlled damage increases TEWL, which is then monitored until values return to baseline. The speed of recovery provides an objective measure of healing capacity without creating open wounds.
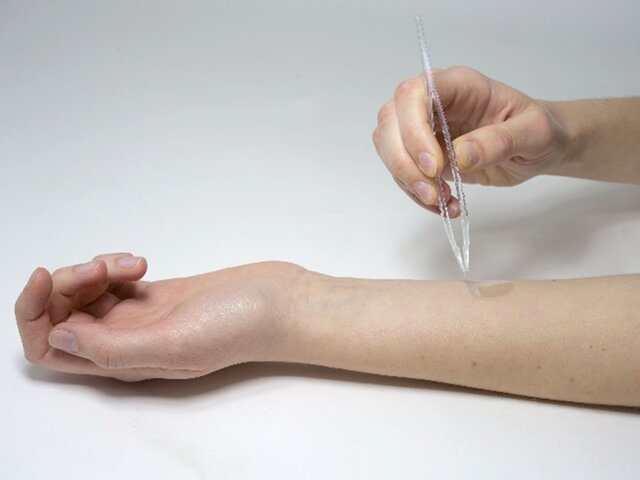
Figure 6. Stripping skin layers by adhesive tapes is a well-recognized way to artificially damage the skin barrier in a standardized way.
Irritation Response
Skin irritation often manifests as redness (Figure 7) (28). Traditional assessment relies on expert visual grading, but human perception struggles with subtle differences, day-to-day comparisons, and the perception of redness for different Fitzpatrick types. Standardized photography provides more consistent evaluation, and added image analysis software enables automated colour quantification (29).
Besides photography, handheld colour measuring probes offer a quick and objective alternative for redness measurement (30).
Other parameters, such as surface topography and elasticity, can add information to provide a broader picture of skin health.
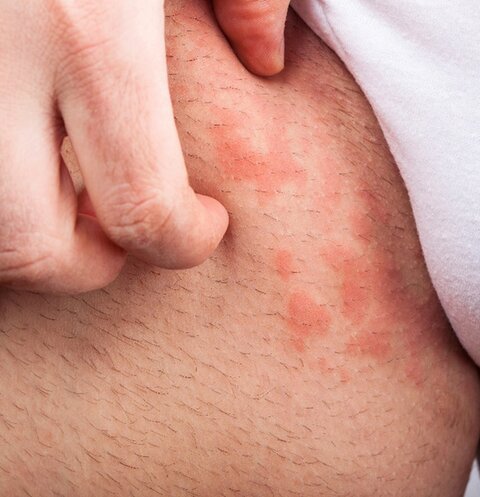
Figure 7. A compromised skin barrier is prone to irritation.
Perspectives and Ongoing Questions
Despite the progress in microbiome research, major challenges remain. No single microbial profile defines healthy skin, and the wide inter- and intra-individual variation makes universal conclusions unlikely. Microbiome analysis alone is therefore insufficient to demonstrate the benefit of microbiome-targeting products.
Traditional skin testing methods, by contrast, remain robust and reliable. They provide measurable outcomes—hydration, TEWL, pH, sebum levels, irritation response—directly linked to skin health. For this reason, they continue to serve as the foundation for substantiating product claims, even when those claims are framed in microbiome-related language.
Although everyone is talking about the microbiome, there are still many unsolved questions. How can we promote the diversity of the microbiome and prevent dysbiosis? What role does the microbiome play in certain diseases, and how can we use it therapeutically? What are the effects of post-, pro- and prebiotics, and cosmetic procedures on microbial communities? How do environmental influences affect the microbiome?
These topics need to be addressed in future tests and might even lead to the definition of new skin types, taking into account their predominant microbial communities. This would fundamentally change how dermatology and cosmetology categorize and treat skin and could even reinforce the already booming trend towards the individualisation of cosmetic products (Figure 8).

Figure 8. The definition of new skin types considering also the predominant microbial communities, requires a stronger specialisation of off-the-shelf products.
Conclusions
The skin microbiome is a fascinating and rapidly evolving research field. However, despite considerable scientific activity, no consensus exists on an “ideal” microbiome. The complexity and variability of microbial communities mean that microbiome analysis, while informative, rarely provides conclusive evidence of product efficacy.
For now, the most practical and scientifically reliable approach remains the use of traditional biophysical skin testing methods and imaging techniques that allow researchers to prove claims about skin health independently of microbiome analysis.The future of microbiome skin care research lies in bridging these two approaches: combining microbiome profiling with established skin health assessments. This integrated perspective will clarify how products influence both the skin’s physical state and its microbial inhabitants, helping to ensure that interventions genuinely support long-term health rather than disrupt delicate balances.
Conclusion
The future of cosmetics lies in the continued evolution of holistic approaches which represents a transformative shift in the industry, merging scientific advancements, natural ingredients, and wellness principles. By understanding and embracing the interconnectedness of these elements, the cosmetics industry can cultivate products that not only enhance external beauty but also contribute to the overall well-being of individuals and the planet.
The interplay between beauty from within and topical cosmetics is the key for future products. The integration of biotechnology and green chemistry is revolutionizing cosmetic formulations, offering sustainable and biocompatible alternatives.
Developers can implement blockchain to trace the journey of ingredients from source to product. Nevertheless, the efficacy of the natural products should be scientifically proven. Marketers can communicate transparency as a brand value, and parallelly educate consumers by highlighting how specific ingredients contribute to radiant and healthy skin.
By embracing the synergy between these approaches and leveraging scientific advancements, the cosmetics industry can provide consumers with comprehensive beauty solutions that cater to both internal and external dimensions of beauty.
Surfactant Applications

The application area lends itself particularly well to the use of AI. Active today in this area is the US company Potion AI (6). The company provides AI-powered formulation tools for beauty and personal care R&D. Their offerings include Potion GPT, next generation ingredient and formula databases and AI document processing. Potion’s work could have a significant impact on the entire surfactant value chain, from raw material suppliers to end consumers. By using their GPT technology, they can help target work toward novel surfactant molecules that have optimal properties for specific applications. By using their ingredient and formula databases, they can access and analyze a vast amount of data on surfactant performance, safety, and sustainability. By using their AI document processing, they can extract and organize relevant information from patents, scientific papers, and regulatory documents. These capabilities could enable Potion AI's customers to design and optimize surfactant formulations that are more effective, eco-friendly, and cost-efficient. A particularly interesting application for this type of capability is deformulation.
Deformulation is the process of reverse engineering a product's formulation by identifying and quantifying its ingredients. Deformulation can be used for various purposes, such as quality control, competitive analysis, patent infringement, or product improvement. However, deformulation can be challenging, time-consuming, and costly, as it requires sophisticated analytical techniques, expert knowledge, and access to large databases of ingredients and formulas.
AI can potentially enhance and simplify the deformulation process by using data-driven methods to infer the composition and structure of a product from its properties and performance. For example, AI can use machine learning to learn the relationships between ingredients and their effects on the product's characteristics, such as color, texture, fragrance, stability, or efficacy. AI can also use natural language processing to extract and analyze information from various sources, such as labels, patents, literature, or online reviews, to identify the possible ingredients and their concentrations in a product.
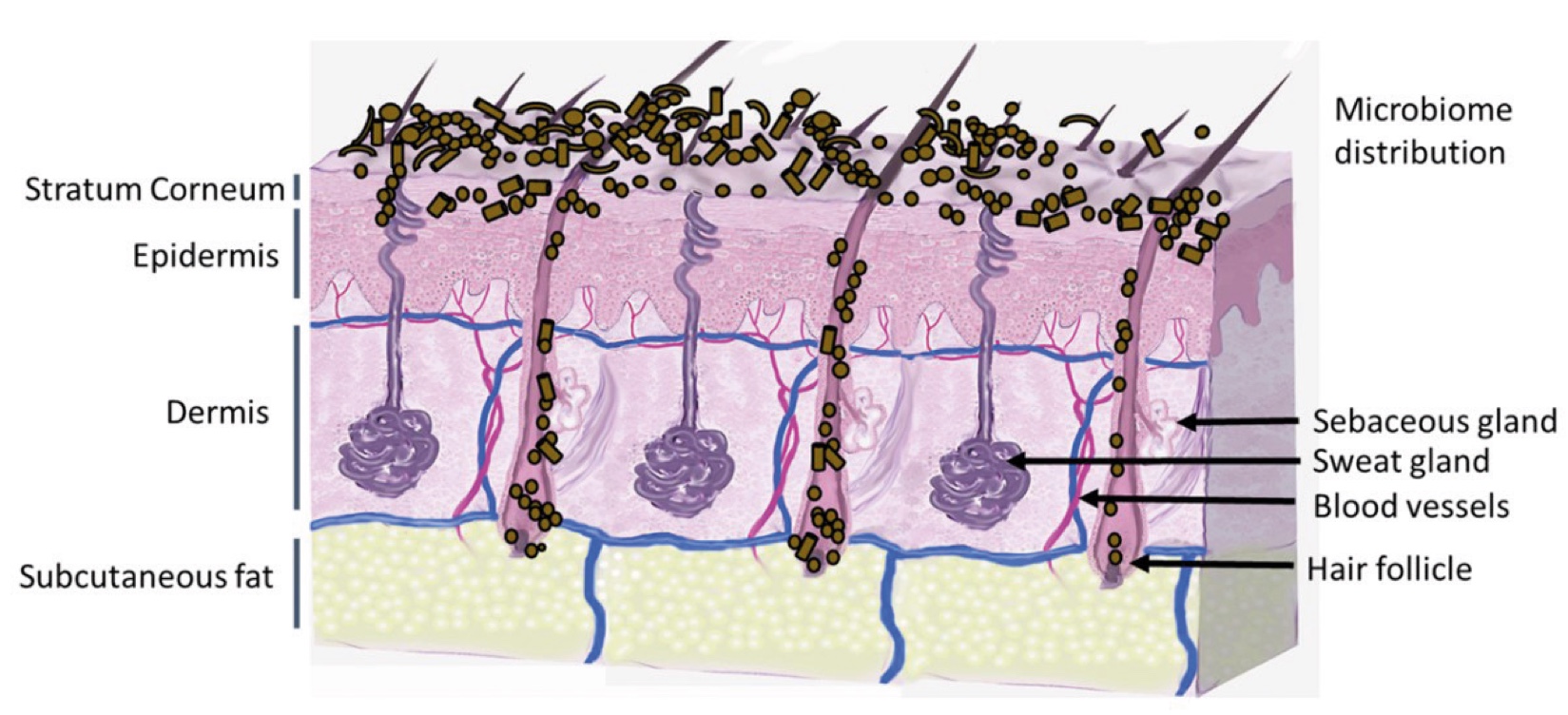
Figure 2. Skin Section with Microbiome. Most microorganisms live in the superficial layers of the stratum corneum and in the upper parts of the hair follicles. Some reside in the deeper areas of the hair follicles and are beyond the reach of ordinary disinfection procedures. There bacteria are a reservoir for recolonization after the surface bacteria are removed.
References and notes
- https://commonfund.nih.gov/hmp
Wang B, Yao M, Lv L, Ling Z, Li L. The Human Microbiota in Health and Disease, Engineering, Volume 3, Issue 1, 2017, Pages 71-82, ISSN 2095-8099, https://doi.org/10.1016/J.ENG.2017.01.008. (https://www.sciencedirect.com/science/article/pii/S2095809917301492 )
Sanford JA, Gallo RL. Functions of the skin microbiota in health and disease. Semin. Immunol. 2013, Nov 30;25(5):370-7. doi: 10.1016/j.smim.2013.09.005. Epub 2013 Nov 20. PMID: 24268438; PMCID: PMC4219649.
Grice EA, Segre, JA. The skin microbiome, Nat. Rev. Microbio.l 9, 244–253 (2011). https://doi.org/10.1038/nrmicro2537
Grice EA, Kong HH, Conlan S, Deming CB, Davis J, Young AC, Bouffard GG, Blakesley RW, Murray PR, Green ED, Turner ML, Segre JA. Topographical and temporal diversity of the human skin microbiome, Science. 2009 May 29;324(5931):1190-2. doi: 10.1126/science.1171700. PMID: 19478181; PMCID: PMC2805064.
Costello EK, Lauber CL, Hamady M, Fierer N, Gordon JI, Knight R. Bacterial community variation in human body habitats across space and time, Science. 2009 Dec 18;326(5960):1694-7. doi: 10.1126/science.1177486. Epub 2009 Nov 5.
Lee HJ, Kim M. Skin Barrier Function and the Microbiome. Int J Mol Sci. 2022 Oct 28;23(21):13071. doi: 10.3390/ijms232113071. PMID: 36361857; PMCID: PMC9654002.
https://eur-lex.europa.eu/legal-content/EN/ALL/?uri=celex%3A32009R1223
- Mukherjee S, Mitra R, Maitra A, Gupta S, Kumaran S, Chakrabortty A, Majumdeet PP. Sebum and Hydration Levels in Specific Regions of Human Face Significantly Predict the Nature and Diversity of Facial Skin Microbiome. Sci Rep6, 36062 (2016). https://doi.org/10.1038/srep36062
Mim MF, Sikder MH, ChowdhuryMZH, Bhuiyan A, Zinan NN, Islam SMN. The dynamic relationship between skin microbiomes and personal care products: A comprehensive review, Heliyon, Volume 10, Issue 14, 2024, e34549, ISSN 2405-8440, https://doi.org/10.1016/j.heliyon.2024.e34549., (https://www.sciencedirect.com/science/article/pii/S2405844024105804 )
van der Hoeven H, Prade H. Healthy skin has healthy microbiota: how can we help?, Personal Care North America, October 2019
Prajapati SK, Lekkala L, Yadav D, Jain S, Yadav H. Microbiome and Postbiotics in Skin Health. Biomedicines. 2025; 13(4):791. https://doi.org/10.3390/biomedicines13040791
Kottner J, Lichterfeld A, Blume-Peytavi U. Transepidermal water loss in young and aged healthy humans: a systematic review and meta-analysis. Arch Dermatol Res. 2013 May;305(4):315-23. doi: 10.1007/s00403-012-1313-6. Epub 2013 Jan 23. PMID: 23341028.
Alexander H, Brown S, Danby S, Flohr C. Research Techniques Made Simple: Transepidermal Water Loss Measurement as a Research Tool. J Invest Dermatol. 2018 Nov;138(11):2295-2300.e1. doi: 10.1016/j.jid.2018.09.001. PMID: 30348333.
Akdeniz M, Gabriel S, Lichterfeld-Kottner A, Blume-Peytavi U, Kottner J. Transepidermal water loss in healthy adults: a systematic review and meta-analysis update. Br J Dermatol. 2018 Nov;179(5):1049-1055. doi: 10.1111/bjd.17025. Epub 2018 Sep 9. PMID: 30022486. (https://onlinelibrary.wiley.com/doi/abs/10.1111/bjd.17025 )
Montero-Vilchez T, Segura-Fernández-Nogueras M-V, Pérez-Rodríguez I, Soler-Gongora M, Martinez-Lopez A, Fernández-González A, Molina-Leyva A, Arias-Santiago S. Skin Barrier Function in Psoriasis and Atopic Dermatitis: Transepidermal Water Loss and Temperature as Useful Tools to Assess Disease Severity. Journal of Clinical Medicine. 2021; 10(2):359. https://doi.org/10.3390/jcm10020359
Osseiran S, Cruz JD, Jeong S, Wang H, Fthenakis C, Evans CL. Characterizing stratum corneum structure, barrier function, and chemical content of human skin with coherent Raman scattering imaging. Biomed Opt Express. 2018 Nov 26;9(12):6425-6443. doi: 10.1364/BOE.9.006425. PMID: 31065440; PMCID: PMC6490993.
Rawlings AV, Matts PJ. Stratum corneum moisturization at the molecular level: an update in relation to the dry skin cycle. J Invest Dermatol. 2005 Jun;124(6):1099-110. doi: 10.1111/j.1523-1747.2005.23726.x. PMID: 15955083. (https://www.sciencedirect.com/science/article/pii/S0022202X15323010 )
- Thune P, Nilsen T, Hanstad IK, Gustavsen T, Lövig Dahl H. The water barrier function of the skin in relation to the water content of stratum corneum, pH and skin lipids. The effect of alkaline soap and syndet on dry skin in elderly, non-atopic patients. Acta Derm Venereol. 1988;68(4):277-83. PMID: 2459871.
Warner RR, Stone KJ, Boissy YL. Hydration disrupts human stratum corneum ultrastructure. J Invest Dermatol. 2003 Feb;120(2):275-84. doi: 10.1046/j.1523-1747.2003.12046.x. PMID: 12542533. (https://www.sciencedirect.com/science/article/pii/S0022202X15301676 )
Charalambopoulou GC, Steriotis TA, Hauss T, Stubos AK, Kanellopoulos NK. Structural alterations of fully hydrated human stratum corneum. Physica B: Condensed Matter. Volume 350, Issues 1–3, Supplement, 15 July 2004, Pages E603-E606 350. 10.1016/j.physb.2004.03.161.
Rippke F, Schreiner V, Schwanitz HJ. The acidic milieu of the horny layer: new findings on the physiology and pathophysiology of skin pH. Am J Clin Dermatol. 2002;3(4):261-72. doi: 10.2165/00128071-200203040-00004. PMID: 12010071.
Sator PG, Schmidt JB, Hönigsmann H. Comparison of epidermal hydration and skin surface lipids in healthy individuals and in patients with atopic dermatitis. J Am Acad Dermatol. 2003 Mar;48(3):352-8. doi: 10.1067/mjd.2003.105. PMID: 12637914. (https://www.sciencedirect.com/science/article/abs/pii/S0190962202615099 )
Clayton, R.W., Göbel, K., Niessen, C.M., Paus, R., Steensel, M.A.M., Lim, X. Homeostasis of the sebaceous gland and mechanisms of acne pathogenesis, British Journal of Dermatology Br J Dermatol, 2019, 181 (4): 677-690, https://doi.org/10.1111/bjd.17981.
Del Rosso JQ, Kircik L. The primary role of sebum in the pathophysiology of acne vulgaris and its therapeutic relevance in acne management. J Dermatolog Treat. 2024 Dec;35(1):2296855. doi: 10.1080/09546634.2023.2296855. Epub 2023 Dec 26. PMID: 38146664.
Dobrev H. Fluorescence diagnostic imaging in patients with acne. Photodermatol Photoimmunol Photomed. 2010 Dec;26(6):285-9. doi: 10.1111/j.1600-0781.2010.00541.x. PMID: 21091785. (https://onlinelibrary.wiley.com/doi/10.1111/j.1600-0781.2010.00541.x )
Lanzendörfer G., Uhl C. How Effective is Your Anti-Acne Product, SPC, December 2018, Volume 91 (12): Pages 64-66
Wahlberg JE, Lindberg M. Assessment of Skin Blood Flow – An Overview, in Bioengineering of the Skin: Cutaneous Blood Flow and Erythema (Berardesca E, Ed.), 1995, Pages 23-27
Frew J, Penzi L, Suarez-Farinas M, Garcet S, Brunner PM, Czarnowicki T, Kim J, Bottomley C, Finney R, Cueto I, Fuentes-Duculan J, Ohmatsu H, Lentini T, Yanofsky V, Krueger JG, Guttman-Yassky E, Gareau D. The erythema Q-score, an imaging biomarker for redness in skin inflammation. Exp Dermatol. 2021 Mar;30(3):377-383. doi: 10.1111/exd.14224. Epub 2020 Nov 30. PMID: 33113259; PMCID: PMC8049083.
Clarys P, Alewaeters K, Lambrecht R, Barel AO. Skin color measurements: comparison between three instruments: the Chromameter(R), the DermaSpectrometer(R) and the Mexameter(R). Skin Research and Technology : Official Journal of International Society for Bioengineering and the Skin (ISBS) [and] International Society for Digital Imaging of Skin (ISDIS) [and] International Society for Skin Imaging (ISSI). 2000 Nov;6(4):230-238. DOI: 10.1034/j.1600-0846.2000.006004230.x. PMID: 11428962.
