COLUMN: SCIENCE FOR FORMULATORS
Qualitative analysis of mixed surfactants systems: Synergism and antagonism
Abstract
Surfactants play a pivotal role in the formulation of cosmetics, contributing to the stability, emulsification, and performance of various personal care products. In this paper, we investigate the interactions and complex formation between common anionic, cationic, nonionic and amphoteric surfactants in binary, secondary, ternary and quaternary systems. The findings will provide valuable insights into the design of novel surfactant-based cosmetic formulations with enhanced properties and performance, contributing to the advancement in their knowledge.
Introduction
Despite the many surfactants available nowadays, mixing anionic and cationic surfactants is not a common practice due to the risk of precipitation, which among other things impair each surfactant’s performance. Nevertheless, interactions between molecules of different charges is not only possible but in some cases generates synergism when combined properly.
Historically, the use of anionic and cationic surfactants in a formulation was a practice that was shunned. Mixed anionic/cationic surfactant systems were evaluated were evaluated back in 1981(1, 2).The notion that stable, clear and foaming aqueous combinations of anionic / cationic surfactants could be made using specific surfactants and specified combinations 2005(3, 4, 5, 6).

Objective
The objective of the present study is to evaluate the interactions between anionic, cationic, nonionic and amphoteric surfactants in ternary systems. By studying the formation of hard and soft complexes, we can tailor surfactant combinations to optimize the stability, foaming properties, and solubilization capacity of cosmetic products.
Systems having two types of surfactant (binary systems) have been studied (1). This article expands to systems having three types of surfactants so-called ternary systems.
Materials
Materials. The following surfactants were used in this study without further purification:
1. Brassicamidopropyl Dimethylamine (Procondition 22, 100% a.m.),
2. Behentrimonium Chloride (Quartamin AB, 80% a.m.),
3. Cocamidopropyl Betaine (Probetaina CAPB, 30% a.m.),
4. Cetrimonium Chloride (Sunquart CT29, 29% a.m.),
5. Decyl Glucoside (DG Surfactant, 50% a.m.),
6. Disodium Lauryl Sulfosuccinate (Cosphactant MES 30, 30% a.m.),
7. Dihydrogenated Tallowoylethyl Hydroxyethylmonium Methosulfate (and) Ditallowoylethyl Hydroxyethylmonium Methosulfate (Tetranyl AT-7590, 90% a.m.),
8. Sodium Hydroxypropylsulfonate
9. Laurylglucoside Crosspolymer (Poly SugaNate 160P NC, 40% a.m.),
10. Sodium Cocoyl Glycinate (Miami SCG, 25%),
11. Sodium Laureth-2 Sulfate (Mathless, 70% a.m.),
12. Sodium Laurylglucosides Hydroxypropylsulfonate (SugaNate 160NC, 40% a.m.).
Abbreviations
CAPB - Cocamidopropyl Betaine
CTC - Cetrimonium Chloride
DLSS - Disodium Lauryl Sulfosuccinate
PSN - Sodium Hydroxypropylsulfonate Laurylglucoside Crosspolymer
SCG - Sodium Cocoyl Glycinate
SLES - Sodium Laureth-2 Sulfate
SN - Sodium Laurylglucosides Hydroxypropylsulfonate
The concentrations presented in the studies were based on each material’s active content as described in their technical data sheets.
Methods
Solutions were prepared at different concentrations of surfactants. Mixing ratios are expressed as weight percentages. The room where all tests were carried out was set at 65% RH and 20±0.1°C.
Appearance
Visual inspection was performed placing surfactant aqueous mixtures in transparent 100mL borosilicate glass beakers, which were then placed in a well illuminated room to check for possible formation of precipitates. Samples were rechecked after 24 hours, in case new precipitates would have appeared.
Viscosity
Viscosity measurements were carried out on a digital Brookfield LVT viscometer with appropriate thermostating (±0.1°C), and using the spindle #64. Three different speeds were used for each sample: 6, 30 and 60 rpm.
Results and discussion
Ternary system: anionic-cationic-amphoteric interactions
Ternary systems of anionic-cationic-amphoteric surfactants were also studied. In the first part SLES was chosen as the anionic surfactant, while CTC and CAPB were the cationic and amphoteric surfactants, respectively. Table 1 shows viscosity and appearance results when mixing all the surfactants at different ratios.
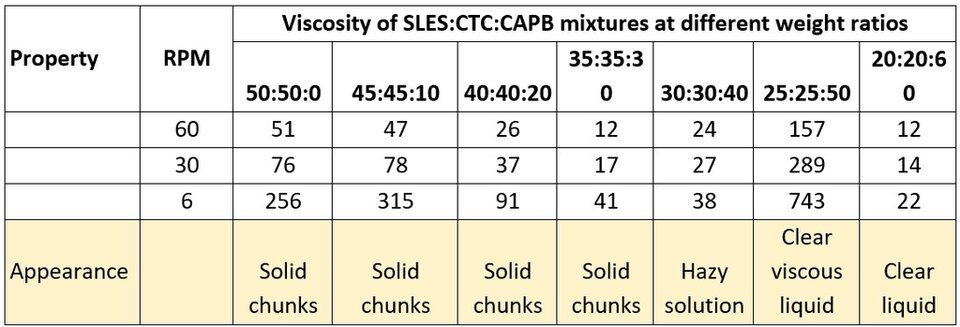
Table 1. Ternary system: anionic-cationic-amphoteric interaction.
It was found that when little or no CAPB was used, precipitates were formed in solutions of mixed surfactants. When the level of CAPB is slightly above that of SLES and CTC, there are no precipitates but a hazy solution. Clear solutions were obtained at two (viscous liquid) and three (liquid solution) times the amount of SLES and CTC used. As discussed in the first part of the study, precipitates formed when mixing SLES and CTC might be due to disordered aggregated microstructures. When adding CAPB, the anionic charge from SLES is partially neutralized by the amine group in CAPB, turning the pair essentially into a nonionic structure, which can then interact with CTC through weaker forces (like ion-dipole or dipole-dipole). It is also possible that the anionic group on CAPB could interact electrostatically with the amine group in CTC, but this interaction might be impaired by the steric conformation of the SLES-CAPB pair. This hypothesis should be confirmed by experimental analysis, as well as for the possible conformational change to worm-like micelles that caused a peak in viscosity, when the amount of CAPB added was two times the amount of both SLES and CTC. When the amount of CAPB is three times the amount of the other two surfactants, a drop in viscosity of the clear solution is observed, which could also correspond to the characteristic scission and branching of previously formed worm-like micelles (7).
The same evaluation was done, replacing SLES by DLSS. The results are shown in Table 2.
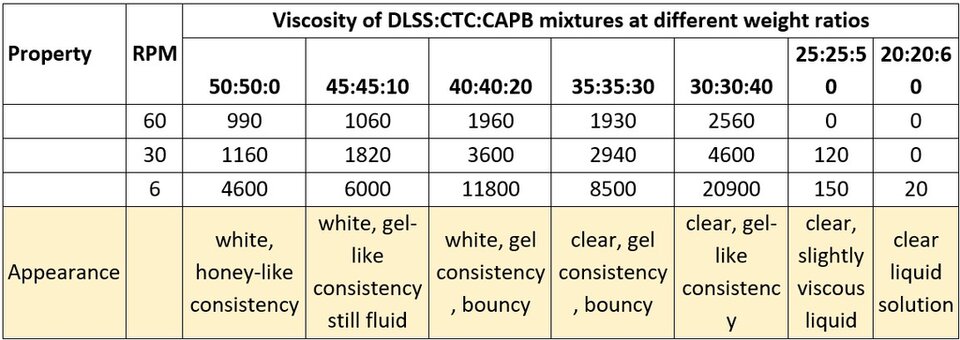
Table 2. Viscosity of DLSS:CTC:CAPB mixtures at different ratio.
Like in the previous test, when little or no CAPB is added to the DLSS-CTC mixture, there are precipitates formed that remain suspended due to the solution's high viscosity. Even more interesting is the fact that the viscosity is high even without CAPB, and with every increase, viscosity increases considerably. When the amount of each surfactant is the same, the mixture gets clear and viscous. The clarity remains during the rest of the ratios tested, but the viscosity peaks when the content of CAPB is approximately 30% higher than the content of both DLSS and CTC.
Another anionic surfactant tested was SCG. The results of that test are shown in Table 3.
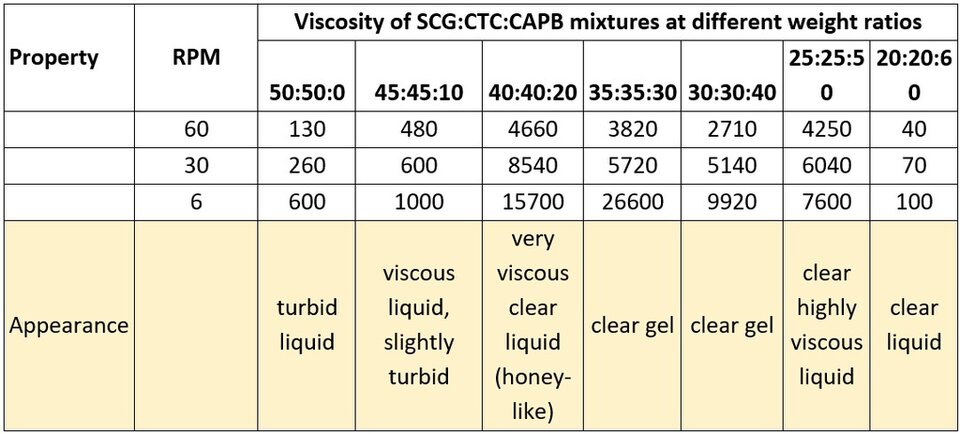
Table 3. Viscosity of SCG:CTC:CAPB mixtures at different ratios.
In the case of SCG, the initial haziness is soon replaced by a clear and viscous liquid when adding CAPB. As a reference, the weight ratio of anionic/cationic/amphoteric surfactant to turn from a hazy mixture into a clear one was 1:1:1.5 for SLES , and 1:1:1 for DLSS. For SCG, the same weight ratio is 1:1:0.5. At higher additions of CAPB, the mixture keeps its clarity, with a peak in viscosity at a weight ratio of around 1:1:1. These results might be explained by the presence of an amide group in SCG, which could increase the solubility of the formed complex, preventing its precipitation. Furthermore, part of SCG structure resembles that of CAPB, which could lead to a better interaction and a possible synergy with very high solution viscosities. Interestingly, increasing levels of CAPB also resulted in a reduction in the solution viscosity, but this reduction is not as marked as with previous anionic surfactants tested. This behavior should be studied more thoroughly and at different pH values, for better understanding.
As in our previous tests, both glucoside anionic surfactants show almost no viscosity response both when mixing them only with CTC, and when CAPB was added after (Tables 4 and 5). This can also be explained by the bulky polar head group present in both surfactants.
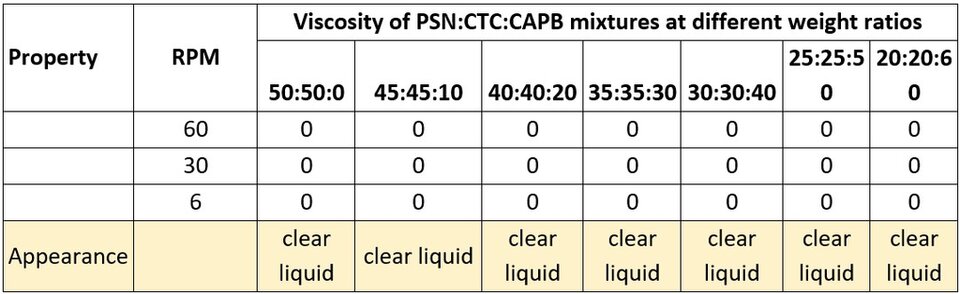
Table 4. Viscosity of PSN:CTC:CAPB mixtures at different ratios.
In the case of SN, a slight increase in viscosity is shown when CAPB is added at half the weight ratio of both SN and CTC (Table 5), but in the context of our study and compared to other viscosity values that we found, that slight increase is not relevant.
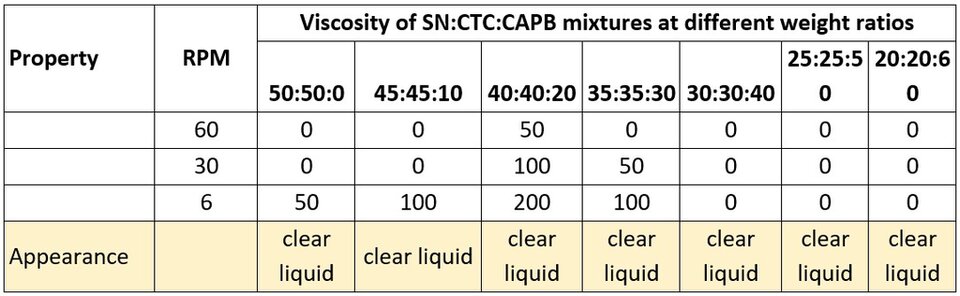
Table 5. Viscosity of SN:CTC:CAPB mixtures at different ratios.
Conclusions
Results from our studies show that synergistic interactions of surfactants are complicated. Properties achieved using specific surfactants at specific concentrations give unexpected results, solubility and viscosity being only two such surprising results (8). We fully expect that additional benefits to such synergistic blends will be discovered that make use of aesthetics and foam alteration.
Acknowledgments
This work would not have been possible without the support of the Department of Chemistry from Pontificia Universidad Catolica del Peru, for which we are grateful.
References and notes
- Rosen MJ, Hua XY (1982) Surface concentrations and molecular interactions in binary mixtures of surfactants. J Colloid Interface Sci 86:164 https://www.semanticscholar.org/paper/Surface-concentrations-and-molecular-interactions-Rosen-Hua/d482fa16cc69e5314175d41d8a68293431e285c4
- Lucassen-Reynders, E. H., Lucassen, J. and Giles, D. (1981). Surface and Bulk Properties of Mixed Anionic/Cationic Surfactant Systems. Journal of Colloid and Interface Science, 81(1), 1-8. https://doi.org/10.1016/0021-9797(81)90312-X
- Rosen MJ, Hua XY (1982) Synergism in binary mixtures of surfactants: II. Some experimental data. J Am Oil Chem Soc 59:582 https://link.springer.com/article/10.1007/BF02636329
- O’Lenick, A. J. (2005). Anionic/Cationic Complexes. Cosmetics & Toiletries, 120(11), 63-64. https://www.scientificspectator.com/documents/surfactant%20spectator/surfactant%20interaction/2005%20Anionic%20Cationic%20Complex.pdf
- O’Lenick, T. (2016). Anionic/cationic interactions. H&PC Today - Household and Personal Care Today, 11(4), 32-35. https://www.teknoscienze.com/Contents/Riviste/Sfogliatore/HPC4_2016/files/assets/basic-html/index.html#34
- Stellner KL, Scamehorn JF (1986) Surfactant precipitation in aqueous solutions containing mixtures of anionic and nonionic surfactants. J Am Oil Chem Soc 63:566 https://beamreach.org/data/091/science/processing/erica/surfactants/nonionic%20surfactants.pdf
- Yin, H., Lin, Y. and Huang, J. (2009). Microstructures and rheological dynamics of viscoelastic solutions in a catanionic surfactant system. Journal of Colloid and Interface Science, 338, 177–183. https://pubmed.ncbi.nlm.nih.gov/19560154/
- Yin, H., Idit, Yli; Y. Inbal, Tzafir, I and Salama Paul (2023) Compatibility Investigation of Cationic Surfactants with Anionic Species Cosmetics 2023, 10(2), 45; https://doi.org/10.3390/cosmetics10020045
