
Regulation
Skin care
peer-reviewed
Ingredients’ claims and how to ensure regulatory compliance while choosing an ingredient
MOJGAN MODDARESI
Founder of Personal Care regulatory and ChemComply (recently joined Registrar Corp), United Kingdom
ABSTRACT:
In the dynamic and ever-evolving world of cosmetics, the language used to describe products carries immense weight. Words have the power to shape perceptions and significantly influence consumer choices. However, with such power comes an equally significant responsibility. The claims made about cosmetic products must be carefully crafted to ensure they are following the regulatory requirement in the EU /UK. In fact, one of the most important responsibilities of the Responsible person based on the regulatory frameworks like Article 20 of the UK and EU Cosmetics Regulations play an important role to put the required criteria in place.
The goal of this article is to clarify the regulations surrounding claims for cosmetic products, ensuring that formulators select ingredients with consideration for the final regulations affecting finished cosmetic products.
The Regulatory Framework: Ensuring Truth and Transparency
??????????????????
“
“A study in healthy women providing probiotic yogurt for four weeks showed an improvement in emotional responses as measured by brain scans”
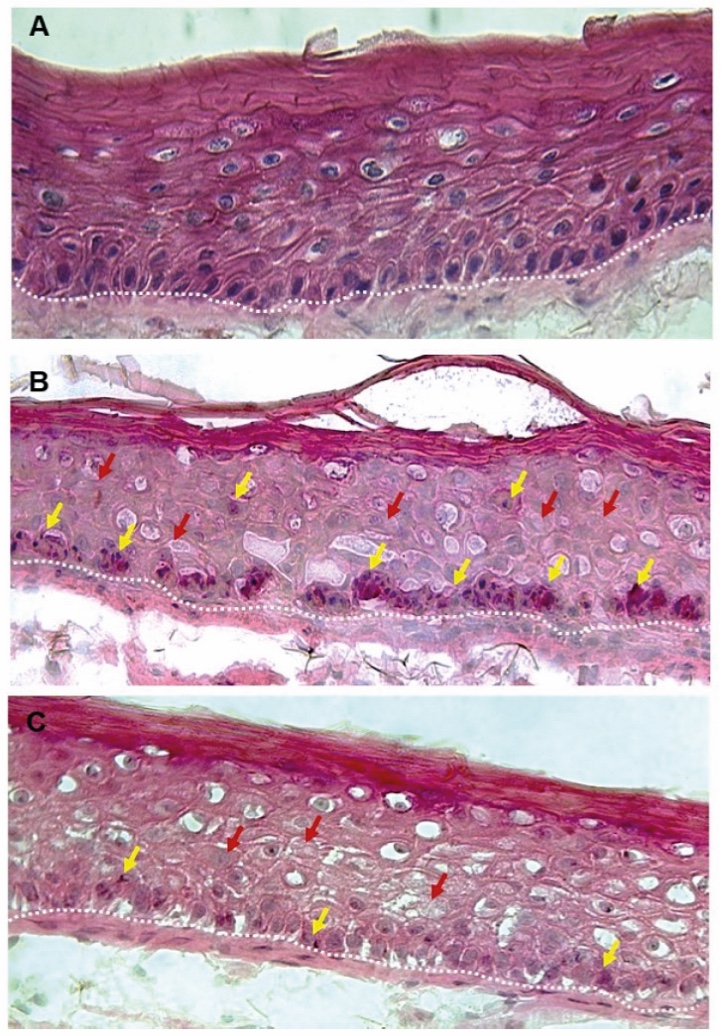
Figure 1. Skin Section with Microbiome. Most microorganisms live in the superficial layers of the stratum corneum and in the upper parts of the hair follicles. Some reside in the deeper areas of the hair follicles and are beyond the reach of ordinary disinfection procedures. There bacteria are a reservoir for recolonization after the surface bacteria are removed.
Materials and methods
Studies of major depressive disorder have been correlated with reduced Lactobacillus and Bifidobacteria and symptom severity has been correlated to changes in Firmicutes, Actinobacteria, and Bacteriodes. Gut microbiota that contain more butyrate producers have been correlated with improved quality of life (1).
A study in healthy women providing probiotic yogurt for four weeks showed an improvement in emotional responses as measured by brain scans (2). A subsequent study by Mohammadi et al. (3) investigated the impacts of probiotic yogurt and probiotic capsules over 6 weeks and found a significant improvement in depression-anxiety-stress scores in subjects taking the specific strains of probiotics contained in the yogurt or capsules. Other studies with probiotics have indicated improvements in depression scores, anxiety, postpartum depression and mood rating in an elderly population (4-7).
Other studies have indicated a benefit of probiotic supplementation in alleviating symptoms of stress. In particular, researchers have looked at stress in students as they prepared for exams, while also evaluating other health indicators such as flu and cold symptoms (1). In healthy people, there is an indication that probiotic supplementation may help to maintain memory function under conditions of acute stress.
Article 20 of both the UK and EU Cosmetics Regulations is a cornerstone in maintaining the integrity of cosmetic claims. It mandates that the language used in labeling, marketing, and advertising must be truthful and not misleading. As consumers become more informed and discerning, the importance of integrity in cosmetic claims has never been more pronounced.
To ensure transparency and consumer protection, the EU Regulation 655/2013 establishes the Common Criteria for Cosmetic Claims. These criteria are referenced in Article 20 and serve as the backbone of ethical marketing in the cosmetics industry. They require all claims to adhere to standards of legal compliance, truthfulness, evidential support, honesty, fairness, and informed decision-making.

The Six Pillars of Ethical Cosmetic Claims
1. Legal Compliance: All claims must comply with existing legal frameworks. This ensures that products are marketed responsibly and in accordance with the law.
2. Truthfulness: Claims must accurately reflect the product's properties and benefits. Misleading or exaggerated claims can erode consumer trust and damage brand reputation.
3. Evidential Support: Claims must be supported by adequate and verifiable evidence. This includes expert assessments and scientific studies that substantiate the product's benefits.
4. Honesty: The language used in claims should be honest and transparent. Consumers should be able to trust that the claims made about a product are genuine and not misleading.
5. Fairness: Claims should be fair and not denigrate competitors. The focus should be on promoting the product's own merits rather than disparaging others.
6. Informed Decision-Making: Consumers should have access to all the information they need to make informed purchasing decisions. This includes clear and accurate labeling and advertising.
Furthermore, the EU commission working group published a technical document in 2017 to provide a tool and clear guideline on Article 20 that can be denigratory This regulatory framework is not just a set of guidelines; it is a commitment to maintaining consumer trust and ensuring that the industry operates with integrity. These criteria are not only obligatory to follow, but it set up a clear guideline to follow specifically for “free from claims”.
According to Article 5 of the EU and UK regulation for the cosmetic products, the responsible person should ensure compliance with Article 20 of the CPR and with the common criteria set out in Commission Regulation (EU) No 655/2013. There is also an important role for the distributors’ channel, and they are responsible to act with duty of the care while interpretating cosmetic claims (Article 6 (1). (1)
Evidential Support: The Backbone of Credible Claims; Extrapolating Ingredients’’ claims to the finished products
One of the most critical aspects of the regulatory framework is the requirement for evidential support. This principle emphasises that claims, whether explicit or implicit, must be backed by adequate and verifiable evidence. This includes expert assessments and scientific studies that substantiate the product's benefits.
A key consideration in this context for choosing the right ingredients’ claim is the differentiation between in vitro and in vivo studies. While in vitro studies can provide valuable insights into ingredient properties, they should not be used to imply the same clinical outcomes as in vivo studies. The challenge lies in extrapolating the properties of ingredients to the final product, ensuring that the claims made are supported by evidence and reflect the product's actual performance.
While designing a product’s efficacy only based on the ingredients’ claims substantiation, it is important to use the exact concentration used in in vivo studies for ingredients. Furthermore the finished products’ claims should be attributed to the ingredients rather than the product itself, while there is no clinical study for the finished products.
Navigating Emerging Trends: The Challenge of Neurocosmetic Claims
One of the most challenging areas for evaluating ingredient claims is the emerging trend of neurocosmetics. These claims focus on improving skin health and mental well-being by influencing the Skin-Brain connection. However, in making such claims, it is essential to adhere to the basic principles of cosmetic definitions.
According to regulatory definitions, a cosmetic product's function is primarily to clean, perfume, change appearance, protect, or correct body odour. Claims that extend beyond these functions, such as those suggesting mood enhancement or stress reduction, can be challenging to substantiate within the regulatory framework. While psychoneurological studies may demonstrate connections between cosmetic ingredients and mood, such claims for the finished products often border on medical definitions, which fall outside the scope of cosmetics. Hence, before getting excited about novel neurocosmetic actives, it is important to consider what would be acceptable claims for the finished products in the framework and definition of cosmetics.
Sustainability Claims: A Rising Trend with Regulatory Challenges
In recent years, sustainability has become a significant focus in the cosmetics industry. Claims around concepts like "eco-friendly," "green," and "kind to the planet" are increasingly popular. However, these environmental claims must be carefully substantiated.
Marketers must ensure that any environmental claims about their products are based on the entire life cycle of the product, from manufacture to disposal. If a general claim cannot be justified, a more limited claim about specific aspects or ingredients may be appropriate. This approach ensures that sustainability claims are truthful and not misleading.
In response to the ongoing climate crisis, in the UK the Advertising Standards Authority (ASA) and the Committee of Advertising Practice (CAP) launched the Climate Change and the Environment (CCE) project in 2021. This initiative aims to ensure that environmental claims in advertising are not misleading or irresponsible, further emphasizing the importance of truthful and substantiated claims in the cosmetics industry (2).
In the EU, Directive (EU) 2024/825 focuses on restricting certain misleading claims on cosmetic artworks, such as: Generic claims such as environmentally friendly, eco-friendly, green, nature's friend, ecological, environmentally correct, climate friendly, gentle on the environment (3).
The EU Commission carried out two inventories of environmental claims looking at 150 environmental claims to evaluate clarity, unambiguity, accuracy and verifiability. The 2020 study found that a considerable share of environmental claims (53.3%) provide vague, misleading or unfounded information about products’ environmental characteristics across the EU and across a wide range of product categories.
Conclusion: Upholding Integrity in a Transformative Industry
As the cosmetics industry continues to evolve, the importance of truthful and substantiated claims cannot be overstated. Regulatory frameworks like Article 20 of the UK and EU Cosmetics Regulations and EU Regulation 655/2013 provide a robust foundation for ethical marketing practices. By adhering to these guidelines, the industry can maintain consumer trust and ensure that products are marketed responsibly.
In a world where consumers are increasingly informed and discerning, the power of words must be wielded with care. By upholding the principles of legal compliance, truthfulness, evidential support, honesty, fairness, and informed decision-making, the cosmetics industry can continue to thrive while maintaining the integrity of its claims. As new trends and challenges emerge, the commitment to transparency and consumer protection remains paramount, ensuring that the industry operates with integrity and responsibilities.
Conclusion
The future of cosmetics lies in the continued evolution of holistic approaches which represents a transformative shift in the industry, merging scientific advancements, natural ingredients, and wellness principles. By understanding and embracing the interconnectedness of these elements, the cosmetics industry can cultivate products that not only enhance external beauty but also contribute to the overall well-being of individuals and the planet.
The interplay between beauty from within and topical cosmetics is the key for future products. The integration of biotechnology and green chemistry is revolutionizing cosmetic formulations, offering sustainable and biocompatible alternatives.
Developers can implement blockchain to trace the journey of ingredients from source to product. Nevertheless, the efficacy of the natural products should be scientifically proven. Marketers can communicate transparency as a brand value, and parallelly educate consumers by highlighting how specific ingredients contribute to radiant and healthy skin.
By embracing the synergy between these approaches and leveraging scientific advancements, the cosmetics industry can provide consumers with comprehensive beauty solutions that cater to both internal and external dimensions of beauty.
Surfactant Applications

The application area lends itself particularly well to the use of AI. Active today in this area is the US company Potion AI (6). The company provides AI-powered formulation tools for beauty and personal care R&D. Their offerings include Potion GPT, next generation ingredient and formula databases and AI document processing. Potion’s work could have a significant impact on the entire surfactant value chain, from raw material suppliers to end consumers. By using their GPT technology, they can help target work toward novel surfactant molecules that have optimal properties for specific applications. By using their ingredient and formula databases, they can access and analyze a vast amount of data on surfactant performance, safety, and sustainability. By using their AI document processing, they can extract and organize relevant information from patents, scientific papers, and regulatory documents. These capabilities could enable Potion AI's customers to design and optimize surfactant formulations that are more effective, eco-friendly, and cost-efficient. A particularly interesting application for this type of capability is deformulation.
Deformulation is the process of reverse engineering a product's formulation by identifying and quantifying its ingredients. Deformulation can be used for various purposes, such as quality control, competitive analysis, patent infringement, or product improvement. However, deformulation can be challenging, time-consuming, and costly, as it requires sophisticated analytical techniques, expert knowledge, and access to large databases of ingredients and formulas.
AI can potentially enhance and simplify the deformulation process by using data-driven methods to infer the composition and structure of a product from its properties and performance. For example, AI can use machine learning to learn the relationships between ingredients and their effects on the product's characteristics, such as color, texture, fragrance, stability, or efficacy. AI can also use natural language processing to extract and analyze information from various sources, such as labels, patents, literature, or online reviews, to identify the possible ingredients and their concentrations in a product.
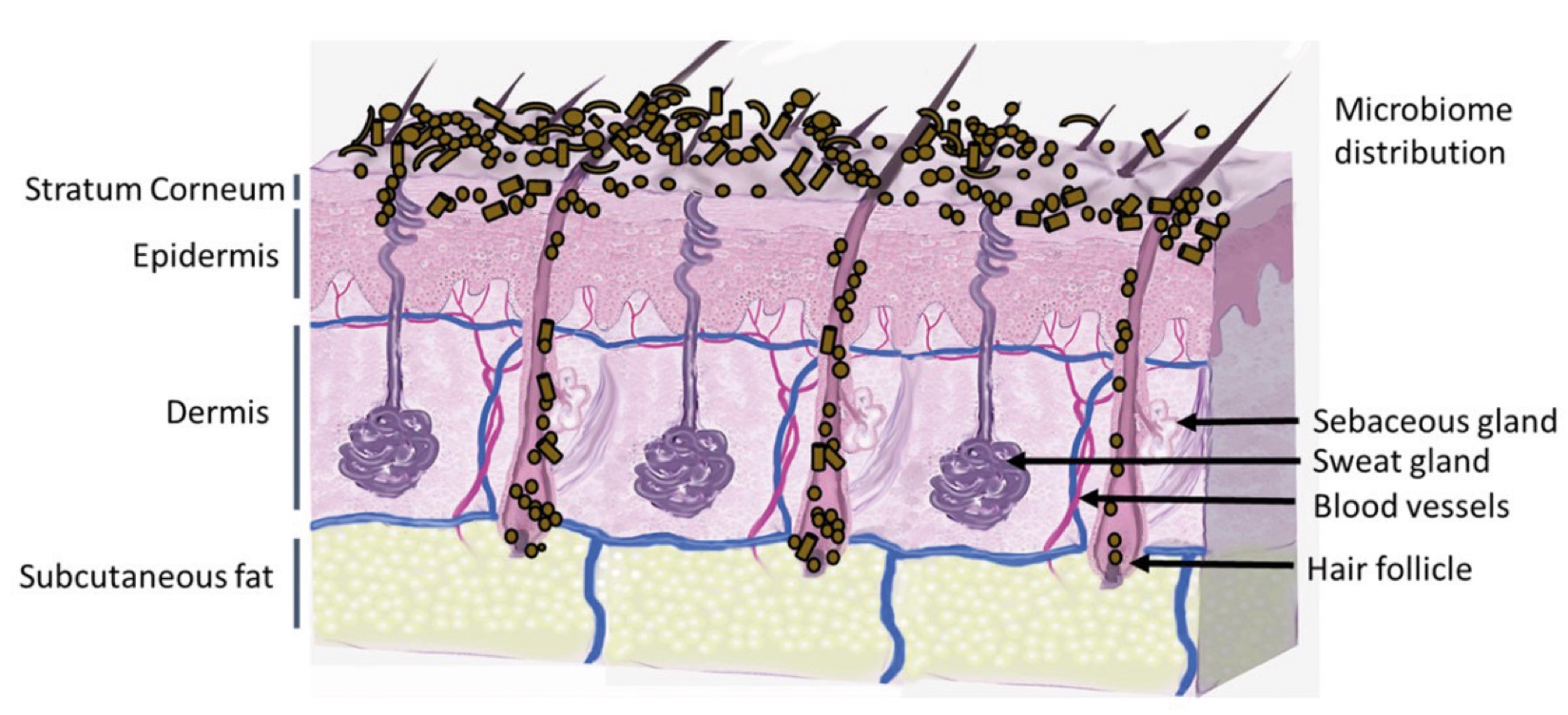
Figure 2. Skin Section with Microbiome. Most microorganisms live in the superficial layers of the stratum corneum and in the upper parts of the hair follicles. Some reside in the deeper areas of the hair follicles and are beyond the reach of ordinary disinfection procedures. There bacteria are a reservoir for recolonization after the surface bacteria are removed.
References and notes
- Technical document on cosmetic claims. Agreed by the Sub-Working Group on Claims (version of 3 July 2017) https://www.google.co.uk/url?sa=t&source=web&rct=j&opi=89978449&url=https://ec.europa.eu/docsroom/documents/24847/attachments/1/translations/en/renditions/native&ved=2ahUKEwi10Yu_yIaLAxWaS0EAHVGyPYYQFnoECBMQAQ&usg=AOvVaw1oitALj_6Y48K02uwLYVnQ
- The environment: misleading claims and social responsibility in advertising. Retrieved from: https://www.asa.org.uk/static/d819e399-3cf9-44ea-942b82d5ecd6dff3/4d3c736f-1e59-471f-bf77e10614544b3b/CAP-guidance-on-misleading-environmental-claims-and-social-responsibility.pdf
- Green claims. New criteria to stop companies from making misleading claims about environmental merits of their products and services. Retrieved from: https://environment.ec.europa.eu/topics/circular-economy/green-claims_en
