
Regulation
Skin care
KEYWORDS
peer-reviewed
The Dermocosmetic Claim Constraint
Olivia Santoni
CEO Bloom Regulatory Ltd, London, England
ABSTRACT: Dermo-cosmetics have become an increasingly popular sector in the cosmetic industry due to combining cosmetic formulation with dermatology-inspired science. While often seen as more advanced, these products remain cosmetics by definition and must comply with the regulatory framework. Given their positioning, a central challenge for companies in this space is navigating the fine line between cosmetic claims and medicinal claims. The 'dermocosmetic' category often pushes the boundaries of acceptable cosmetic claims, focusing on mechanisms of action and efficacy that approach a therapeutic effect. This article offers a summary of the primary challenges and provides guidance to ensure adherence to the relevant legislation.
??????????????????
“
“A study in healthy women providing probiotic yogurt for four weeks showed an improvement in emotional responses as measured by brain scans”
Figure 1. Skin Section with Microbiome. Most microorganisms live in the superficial layers of the stratum corneum and in the upper parts of the hair follicles. Some reside in the deeper areas of the hair follicles and are beyond the reach of ordinary disinfection procedures. There bacteria are a reservoir for recolonization after the surface bacteria are removed.
Materials and methods
Studies of major depressive disorder have been correlated with reduced Lactobacillus and Bifidobacteria and symptom severity has been correlated to changes in Firmicutes, Actinobacteria, and Bacteriodes. Gut microbiota that contain more butyrate producers have been correlated with improved quality of life (1).
A study in healthy women providing probiotic yogurt for four weeks showed an improvement in emotional responses as measured by brain scans (2). A subsequent study by Mohammadi et al. (3) investigated the impacts of probiotic yogurt and probiotic capsules over 6 weeks and found a significant improvement in depression-anxiety-stress scores in subjects taking the specific strains of probiotics contained in the yogurt or capsules. Other studies with probiotics have indicated improvements in depression scores, anxiety, postpartum depression and mood rating in an elderly population (4-7).
Other studies have indicated a benefit of probiotic supplementation in alleviating symptoms of stress. In particular, researchers have looked at stress in students as they prepared for exams, while also evaluating other health indicators such as flu and cold symptoms (1). In healthy people, there is an indication that probiotic supplementation may help to maintain memory function under conditions of acute stress.
Introduction
Cosmetics brands and ingredients suppliers have to navigate a complex regulatory landscape to ensure their products remain within the cosmetic definition while still attracting consumers looking to address skin problems. This is an area that can be frustrating to businesses as although robust scientific data may exist to show that an ingredient or a product has a skin health benefit, it is not acceptable to translate those data into therapeutic claims. The main issue we are seeing often is not a lack of data but a regulatory claim constraint.
Definition of Dermocosmetic
The term "dermocosmetic" itself is not legally defined in EU cosmetic legislation (Regulation (EC) No 1223/2009). Instead, it is a marketing term used to position products that bridge the gap between traditional beauty and dermatological science. Companies often use claims focusing on the biological mechanism of action to convey efficacy, which can sometimes stray too close to the boundary of medicinal function. These products typically appeal to consumers dealing with specific skin problems such as for example skin sensitivity, blemish-prone skin or redness-prone skin.
The regulatory challenge is fundamentally defined by the difference between a cosmetic claim and a medicinal/therapeutic claim.
- Cosmetic claims must relate solely to the external parts of the human body (epidermis, hair system, nails, lips, external genital organs) or the teeth and oral mucous membranes, with the exclusive or principal function of cleaning, perfuming, changing appearance, protecting, keeping in good condition, or correcting body odours (1).
- Medicinal claims refer to preventing, diagnosing, treating, or alleviating a disease, or modifying physiological functions by pharmacological, immunological, or metabolic action (2).
Dermo-cosmetics are distinguished by strategically formulated products that prioritise skin health, often incorporating active ingredients supported by strong evidence. These products typically undergo evaluation, including testing under dermatological supervision. Consequently, consumers view dermo-cosmetics, with their science-led formulations and emphasis on higher-quality ingredients, as the optimal choice for promoting skin health.
Challenges - Ingredients Pushing the Boundary
Dermocosmetics is an area of opportunity for both cosmetics brands and ingredients suppliers. Cosmetics brands are used to navigating the cosmetics regulatory framework and will require their suppliers to adhere to the legislation. Therefore ingredient suppliers should always conduct a formal regulatory borderline risk assessment before launching an ingredient concept with a "dermocosmetic" focus. This includes:
- Reviewing the ingredient's classification under EU medicinal product law.
- Evaluating existing product claims using the ingredient to ensure consistency with cosmetic definitions across various Member States.
- Maintaining clear separation between the ingredient's pure scientific/biological data (which may be therapeutic in nature) and the marketing claims designed for the cosmetic market.
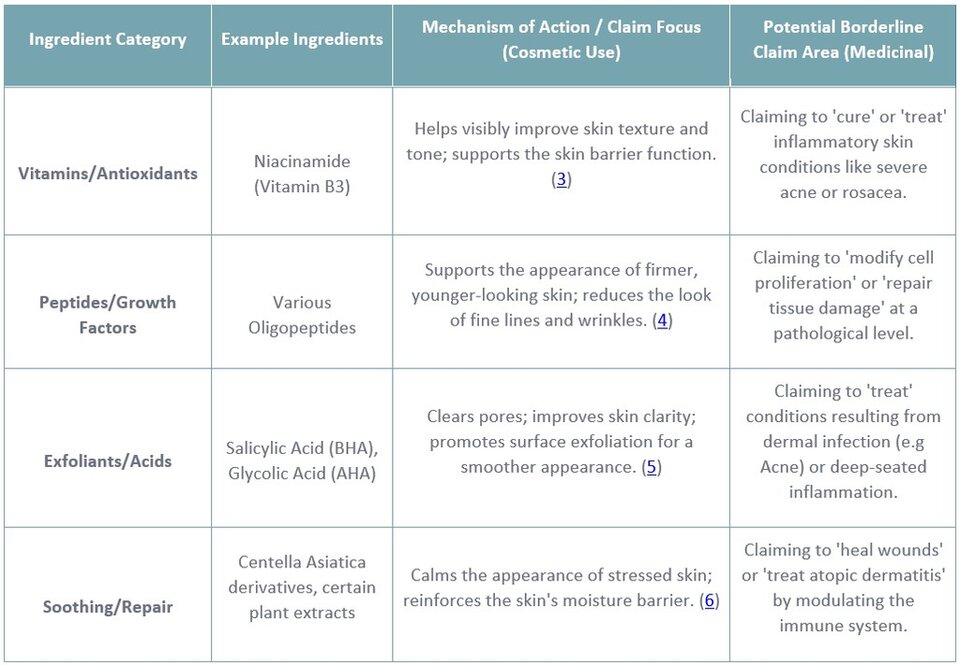
Certain ingredients, due to their established biological activity, are frequently at the centre of this regulatory tension, as their mechanisms of action can suggest a therapeutic effect even when used in a cosmetic context.
Examples include:
Ensuring Compliance: Data Generation and Interpretation Preparation
Compliance should be addressed very early in the development stage to avoid unnecessary cost and the risk of being unable to use data. Careful planning, particularly regarding data generation and interpretation, is therefore crucial. Raw material suppliers and cosmetics brands should consider the following points to ensure success and compliance:
Compliance should be addressed very early in the development stage to avoid unnecessary cost and the risk of being unable to use data. Careful planning, particularly regarding data generation and interpretation, is therefore crucial. Raw material suppliers and cosmetics brands should consider the following points to ensure success and compliance:
- Focus on Cosmetic Endpoints: All efficacy data (including in vitro and in vivo studies) must be focused on demonstrable cosmetic benefits—improvement in appearance, condition, or protection—rather than treating a disease state.
- Avoid Therapeutic Language: Data reports and subsequent marketing summaries for the ingredient must strictly avoid any language suggesting pharmacological action, prevention, or treatment of pathology. Terms like "anti-inflammatory," "cure," "repair," or "therapeutic" should be replaced with cosmetic-appropriate terms like "helps sooth," "comforting," "barrier supporting," or "improving the visible condition of."
Mechanism of Action: While the scientific basis of a mechanism of action may be biological, the final claim must always translate this action into a cosmetic effect. For instance, describing how an ingredient interacts with a cellular pathway is acceptable, but only if the ultimate benefit claimed is an improvement in skin appearance (e.g., reducing visible redness) and not the alleviation of a disease (e.g., treating rosacea).
Conclusion
Although the term "dermocosmetic" is a marketing concept rather than a legal one, the products positioned under this umbrella remain subject to strict cosmetic regulations. The key challenge for brands and ingredient suppliers is navigating the regulatory fine line: translating advanced scientific data—which often details a biological or physiological mechanism of action—into permissible cosmetic claims that focus solely on the appearance and condition of the skin, rather than the treatment or prevention of a disease. Success in the dermocosmetic space hinges on proactive regulatory risk assessment, particularly when developing ingredient concepts. By ensuring that all efficacy data is framed around cosmetic endpoints, and by substituting therapeutic language with legally compliant terminology, companies can leverage the benefits of dermatology-inspired science while maintaining adherence to the cosmetic regulatory framework.
Conclusion
The future of cosmetics lies in the continued evolution of holistic approaches which represents a transformative shift in the industry, merging scientific advancements, natural ingredients, and wellness principles. By understanding and embracing the interconnectedness of these elements, the cosmetics industry can cultivate products that not only enhance external beauty but also contribute to the overall well-being of individuals and the planet.
The interplay between beauty from within and topical cosmetics is the key for future products. The integration of biotechnology and green chemistry is revolutionizing cosmetic formulations, offering sustainable and biocompatible alternatives.
Developers can implement blockchain to trace the journey of ingredients from source to product. Nevertheless, the efficacy of the natural products should be scientifically proven. Marketers can communicate transparency as a brand value, and parallelly educate consumers by highlighting how specific ingredients contribute to radiant and healthy skin.
By embracing the synergy between these approaches and leveraging scientific advancements, the cosmetics industry can provide consumers with comprehensive beauty solutions that cater to both internal and external dimensions of beauty.
Surfactant Applications

The application area lends itself particularly well to the use of AI. Active today in this area is the US company Potion AI (6). The company provides AI-powered formulation tools for beauty and personal care R&D. Their offerings include Potion GPT, next generation ingredient and formula databases and AI document processing. Potion’s work could have a significant impact on the entire surfactant value chain, from raw material suppliers to end consumers. By using their GPT technology, they can help target work toward novel surfactant molecules that have optimal properties for specific applications. By using their ingredient and formula databases, they can access and analyze a vast amount of data on surfactant performance, safety, and sustainability. By using their AI document processing, they can extract and organize relevant information from patents, scientific papers, and regulatory documents. These capabilities could enable Potion AI's customers to design and optimize surfactant formulations that are more effective, eco-friendly, and cost-efficient. A particularly interesting application for this type of capability is deformulation.
Deformulation is the process of reverse engineering a product's formulation by identifying and quantifying its ingredients. Deformulation can be used for various purposes, such as quality control, competitive analysis, patent infringement, or product improvement. However, deformulation can be challenging, time-consuming, and costly, as it requires sophisticated analytical techniques, expert knowledge, and access to large databases of ingredients and formulas.
AI can potentially enhance and simplify the deformulation process by using data-driven methods to infer the composition and structure of a product from its properties and performance. For example, AI can use machine learning to learn the relationships between ingredients and their effects on the product's characteristics, such as color, texture, fragrance, stability, or efficacy. AI can also use natural language processing to extract and analyze information from various sources, such as labels, patents, literature, or online reviews, to identify the possible ingredients and their concentrations in a product.
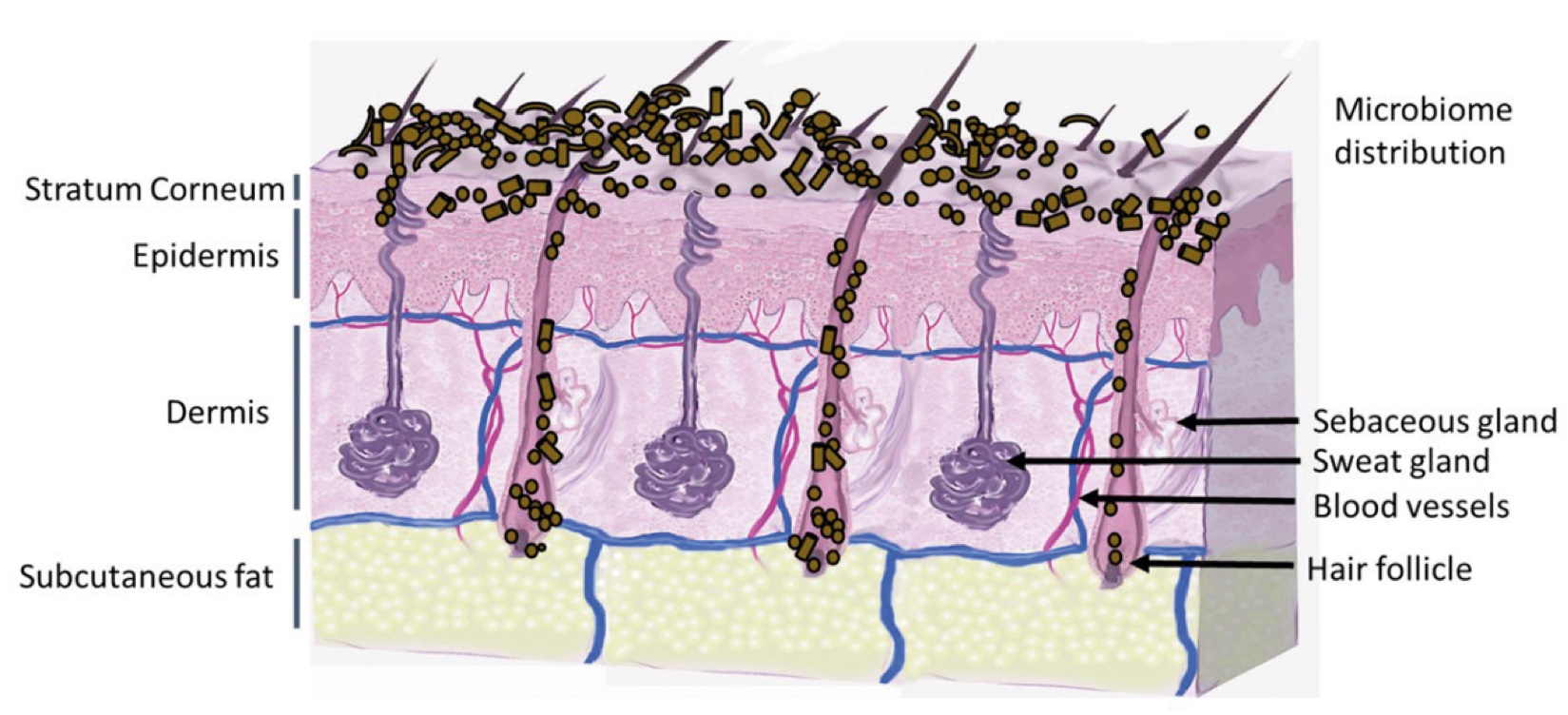
Figure 2. Skin Section with Microbiome. Most microorganisms live in the superficial layers of the stratum corneum and in the upper parts of the hair follicles. Some reside in the deeper areas of the hair follicles and are beyond the reach of ordinary disinfection procedures. There bacteria are a reservoir for recolonization after the surface bacteria are removed.
References and notes
Regulation - 1223/2009 - EN - Cosmetic Products Regulation - EUR-Lex https://eur-lex.europa.eu/eli/reg/2009/1223/oj/eng
Directive - 2001/83 - EN - EUR-Lehttps://eur-lex.europa.eu/eli/dir/2001/83/oj/eng
Lain T., Farris P., Niacinamide: A Multi-functional Cosmeceutical Ingredient. Available from: https://practicaldermatology.com/topics/aesthetics-cosmeceuticals/niacinamide-a-multi-functional-cosmeceutical-ingredient/23720/
Waszkielewicz, A.M.; Mirosław, K. Peptides and Their Mechanisms of Action in the Skin. Appl. Sci. 2024, 14, 11495. https://doi.org/10.3390/app142411495
Jeyaraman B, Kunjaram G, Thomas J, M AJA. A comparative study of 30% salicylic acid peel and 50% glycolic acid peel in mild to moderate acne vulgaris. IP Indian Journal of Clinical and Experimental Dermatology [Internet]. 2025 Feb 8;11(1):121–5. Available from: https://ijced.org/archive/volume/11/issue/1/article/8008#:~:text=Salicylic%20acid%2C%20a%20beta%2Dhydroxy,targeting%20comedones%20and%20inflammatory%20lesions.
Ko K, et al. Enhanced Skin-Protective Effects of a Novel Centella asiatica Variety (BT-Care) Cultivated for 75 Days via Modulation of Antioxidant Defense, Collagen Synthesis, and Skin Barrier Function. Journal of Microbiology and Biotechnology [Internet]. 2025 Jul 14;35:e2504036. Available
